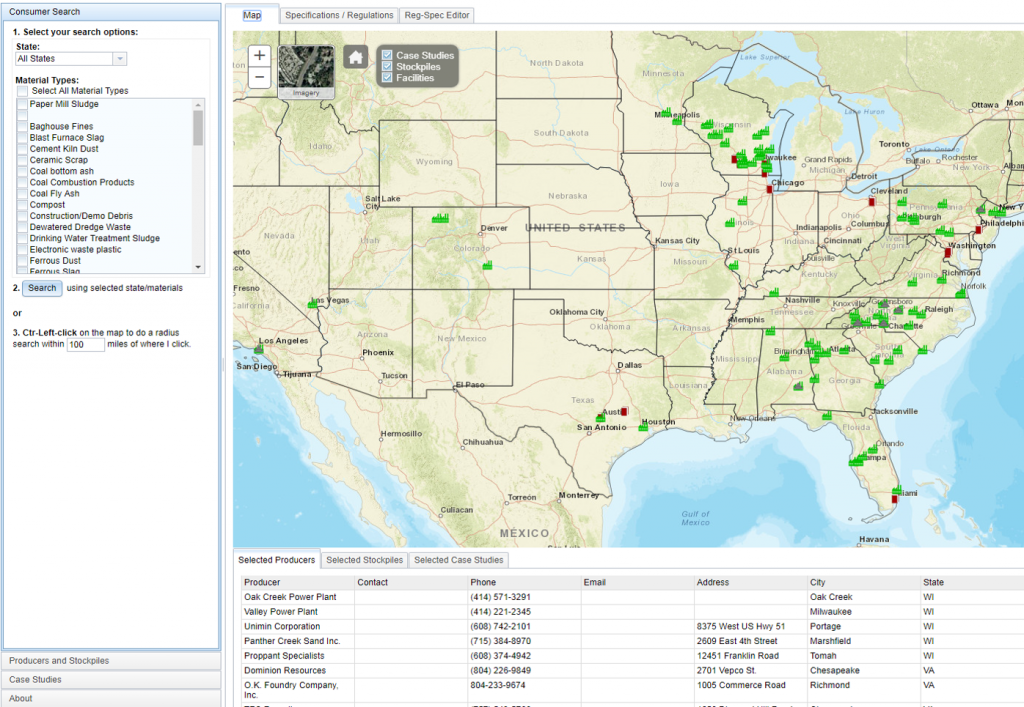
The Recycled Materials Web Map (RMWM) is an on-line Geographic Information System (GIS) web application that connects producers and consumers of recyclable material was developed to assist engineers and contractors in the beneficial reuse of nonhazardous recycled materials in transportation projects. The RMWM is comprised of four core layers: providers, stockpiles, specifications, and case studies. Providers of recycled material can locate their facility and enter contact information. The stockpile layer, connected to the provider layer, allows facility managers to add or update information about their recycled material stockpiles including material type(s), application(s), availability, and cost information. Multiple stockpiles can be associated with each provider. The specification layer includes both Department of Transportation (DOT) specifications and environmental regulations pertaining to the beneficial reuse of recycled material based on specific location, material type, and application. The case study layer locates projects that successfully utilized recycled materials and includes information regarding the material type, application, volume data, and any additional documentation. The web map utilizes search capabilities to locate nearby stockpiles to minimize transportation costs that typically dictate the use of large volumes of materials. The RMWM provides key information which engineers and contractors need to successfully utilize recycled materials, thereby preserving limited natural resources and benefiting the project and society as a whole.
The RMWM is hosted at the Center for Advanced Public Safety (CAPS) at The University of Alabama and was funded through a pooled fund supporting the Recycled Materials Resource Center (RMRC) at the University of Wisconsin-Madison. During the first phase of this work, the RMWM was designed, developed, and tested with preliminary data. A beta version of the web application has been demonstrated to be a useful tool, but there are several areas of the application that need to be addressed for widespread use of this web tool. To address these areas, The University of Alabama has been contracted for a second phase of the project which will wrap up in 2019.